AP Chemistry Topic 8.4 - Acid Base Reactions and Buffers - updated 2026
AP Chemistry Topic 8.4: Acid-Base Reactions and Buffers (Nora Walsh)
This note is a comprehensive summary of Nora Walsh's AP Chemistry video on Topic 8.4, covering acid-base reactions and buffers, including the associated calculations and equation writing for various combinations of strong and weak acids and bases.
---
1. Summary
This video provides a thorough guide to understanding and calculating the results of acid-base reactions, particularly focusing on mixtures. It covers reactions between strong acids and strong bases, weak acids and strong bases, and strong acids and weak bases. The core concepts of neutralization, stoichiometry, and the formation of buffer solutions are explained, along with the necessary calculations to determine pH and the identities of species present after the reaction. The importance of considering the strengths of the acids and bases involved is emphasized throughout.
---
2. Key Takeaways
* **Neutralization:** The reaction between an acid and a base, often forming salt and water. The outcome (pH) depends on the strengths of the reacting species.
* **Strong Acid + Strong Base:** Complete neutralization, resulting in a neutral solution (pH = 7 at 25°C) unless one reactant is in excess.
* **Weak Acid + Strong Base:** The strong base will deprotonate the weak acid, forming its conjugate base. The solution will be basic due to the hydrolysis of the conjugate base. Calculations involve stoichiometry followed by ICE tables for the weak base equilibrium.
* **Strong Acid + Weak Base:** The strong acid will protonate the weak base, forming its conjugate acid. The solution will be acidic due to the hydrolysis of the conjugate acid. Calculations involve stoichiometry followed by ICE tables for the weak acid equilibrium.
* **Buffer Solutions:** Formed when a weak acid and its conjugate base (or a weak base and its conjugate acid) are present in significant amounts. Buffers resist changes in pH upon addition of small amounts of strong acid or base.
* **Henderson-Hasselbalch Equation:** A key tool for calculating the pH of buffer solutions: pH = pKa + log([A-]/[HA]).
* **Before Neutralization (Stoichiometry):** Determine limiting reactants and moles of reactants remaining.
* **After Neutralization (Equilibrium):** If a buffer is formed or if a weak acid/base remains, an ICE table is used to determine equilibrium concentrations and thus pH.
* **Titration Curves:** Visual representations of pH changes during a titration, with distinct shapes for different acid-base combinations.
---
3. Detailed Notes
#### 3.1. Introduction to Acid-Base Reactions
* **Definition:** Reactions between acids and bases.
* **Types of Acids/Bases:**
* **Strong:** Completely ionize/dissociate in water (e.g., HCl, NaOH).
* **Weak:** Partially ionize/dissociate in water (e.g., HF, NH3).
* **Neutralization:** The core process where H+ from an acid reacts with OH- from a base.
* *General Equation:* Acid + Base → Salt + Water
* **Importance of Strengths:** The pH of the final solution depends on whether the acid and base are strong or weak.
#### 3.2. Strong Acid + Strong Base Reactions
* **Example:** HCl (strong acid) + NaOH (strong base) → NaCl (salt) + H2O (water)
* **Net Ionic Equation:** H+ (aq) + OH- (aq) → H2O (l)
* **Outcome:**
* **Equimolar amounts:** Complete neutralization, resulting in a neutral solution (pH = 7 at 25°C).
* **Excess strong acid:** The solution will be acidic. Calculate [H+] from the excess strong acid.
* **Excess strong base:** The solution will be basic. Calculate [OH-] from the excess strong base.
* **Calculations:** Primarily stoichiometric. Use moles of acid and base to determine the limiting reactant and any excess.
#### 3.3. Weak Acid + Strong Base Reactions
* **Example:** HF (weak acid) + NaOH (strong base) → NaF (salt) + H2O (water)
* **Initial Reaction (Stoichiometry):** The strong base will completely react with the weak acid.
* *Equation:* HF + OH- → F- + H2O
* **After Stoichiometry:**
* If the weak acid is completely consumed: The solution will contain the conjugate base of the weak acid (e.g., F-).
* If there is excess weak acid: The solution will contain both the weak acid and its conjugate base.
* **Outcome:**
* **If weak acid is limiting and strong base is in excess:** The dominant species after reaction is excess OH-. The solution is basic.
* **If strong base is limiting and weak acid is in excess:** The dominant species after reaction is the weak acid and its conjugate base (NaF forms F-). This creates a **buffer solution**.
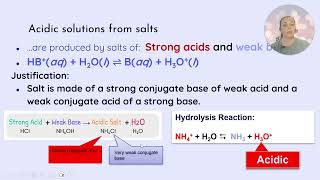
* **If equimolar amounts:** The dominant species after reaction is the conjugate base (e.g., F-). This conjugate base will undergo hydrolysis, making the solution basic.
* **Calculations:**
1. **Stoichiometry:** Determine moles of weak acid and strong base reacted, and calculate moles of weak acid remaining and conjugate base formed.
2. **Equilibrium (ICE Table):** If a buffer is formed or if only the conjugate base remains (and it's not a negligible dissociation), set up an ICE table for the hydrolysis of the conjugate base (or the dissociation of the weak acid if it's in excess and significant).
* For conjugate base hydrolysis (e.g., F- + H2O ⇌ HF + OH-): Use Kb of F-.
* If dealing with a buffer (HA + A-), it's often easier to use the Henderson-Hasselbalch equation.
#### 3.4. Strong Acid + Weak Base Reactions
* **Example:** HCl (strong acid) + NH3 (weak base) → NH4Cl (salt)
* **Initial Reaction (Stoichiometry):** The strong acid will completely react with the weak base.
* *Equation:* H+ + NH3 → NH4+
* **After Stoichiometry:**
* If the weak base is completely consumed: The solution will contain the conjugate acid of the weak base (e.g., NH4+).
* If there is excess weak base: The solution will contain both the weak base and its conjugate acid.
* **Outcome:**
* **If weak base is limiting and strong acid is in excess:** The dominant species after reaction is excess H+. The solution is acidic.
* **If strong acid is limiting and weak base is in excess:** The dominant species after reaction is the weak base and its conjugate acid (NH4Cl forms NH4+). This creates a **buffer solution**.
* **If equimolar amounts:** The dominant species after reaction is the conjugate acid (e.g., NH4+). This conjugate acid will undergo hydrolysis, making the solution acidic.
* **Calculations:**
1. **Stoichiometry:** Determine moles of strong acid and weak base reacted, and calculate moles of weak base remaining and conjugate acid formed.
2. **Equilibrium (ICE Table):** If a buffer is formed or if only the conjugate acid remains (and it's not a negligible dissociation), set up an ICE table for the hydrolysis of the conjugate acid (or the dissociation of the weak base if it's in excess and significant).
* For conjugate acid hydrolysis (e.g., NH4+ + H2O ⇌ NH3 + H3O+): Use Ka of NH4+.
* If dealing with a buffer (B + BH+), it's often easier to use the Henderson-Hasselbalch equation.
#### 3.5. Buffer Solutions
* **Definition:** A solution containing a weak acid and its conjugate base (or a weak base and its conjugate acid) in significant concentrations.
* **Function:** Resists large changes in pH upon the addition of small amounts of strong acid or base.
* **How Buffers Work:**
* **Addition of Strong Acid (H+):** The conjugate base (A-) reacts with the added H+ to form the weak acid (HA).
* *Reaction:* A- + H+ → HA
* **Addition of Strong Base (OH-):** The weak acid (HA) reacts with the added OH- to form the conjugate base (A-) and water.
* *Reaction:* HA + OH- → A- + H2O
* **Henderson-Hasselbalch Equation (for weak acid/conjugate base buffers):**
* **pH = pKa + log ([A-]/[HA])**
* Where:
* pKa = -log(Ka)
* [A-] = concentration of the conjugate base
* [HA] = concentration of the weak acid
* **For weak base/conjugate acid buffers:**
* Use pKb of the weak base.
* The equation can be written as: **pOH = pKb + log ([BH+]/[B])** (where [BH+] is the conjugate acid and [B] is the weak base).
* Alternatively, convert to Ka of the conjugate acid and use the standard Henderson-Hasselbalch equation. Remember that Ka * Kb = Kw, so pKa + pKb = 14 (at 25°C).
* **When to Use:**
* When both the weak acid and its conjugate base are present in significant amounts (usually from a weak acid + strong base reaction where the base is limiting, or vice-versa).
* When calculating the pH of a solution that *is* a buffer.
#### 3.6. General Calculation Strategy for Mixtures
1. **Identify all species present:** Write out the formulas for the acid(s) and base(s).
2. **Determine strengths:** Classify each as strong or weak.
3. **Write the reaction equation(s):** Focus on the reaction between the strongest acid and strongest base present.
* Strong acid + Strong base → Salt + Water (complete neutralization)
* Strong acid + Weak base → Conjugate acid of weak base (complete reaction with strong acid)
* Weak acid + Strong base → Conjugate base of weak acid (complete reaction with strong base)
* Weak acid + Weak base → Usually negligible reaction, but can react to form conjugate species.
4. **Perform Stoichiometric Calculations:** Use moles to determine the limiting reactant and the amounts of species remaining after the reaction.
5. **Analyze the Remaining Species:**
* **Excess Strong Acid/Base:** Calculate pH/pOH directly from the concentration of the excess.
* **Buffer System:** If a weak acid and its conjugate base (or vice-versa) are present in significant amounts, use the Henderson-Hasselbalch equation.
* **Only Weak Acid/Base or Conjugate Species:** If a weak acid or its conjugate base (or a weak base or its conjugate acid) remains, and it's not part of a buffer, set up an ICE table to determine the equilibrium concentrations and pH/pOH.
#### 3.7. Titration Curves (Brief Mention)
* The video touches on how these reactions manifest in titration curves. The shape of the curve (initial pH, midpoint, equivalence point, final pH) is characteristic of the strengths of the acid and base being titrated. This is a related concept where these reaction calculations are applied.
---
Related Summaries
Why this video matters
This video provides valuable insights into the topic. Our AI summary attempts to capture the core message, but for the full nuance and context, we highly recommend watching the original video from the creator.
Disclaimer: This content is an AI-generated summary of a public YouTube video. The views and opinions expressed in the original video belong to the content creator. YouTube Note is not affiliated with the video creator or YouTube.
![[캡컷PC]0015-복합클립만들기분리된영상 하나로 만들기](https://img.youtube.com/vi/qtUfil0xjCs/mqdefault.jpg)
